When you perform an internal audit, you will be able to compare your quality management system to the requirements and understand if there are any non-conformances. Internal audits will be used to assess conformity, evaluate effectiveness and identify opportunities for improvement. (We offer internal audit training to ensure your internal auditors are able to perform an effective internal audit as well as an audit checklist to help guide your internal auditors on covering all areas of your QMS.) Internal audits are an ISO 13485 requirement and they are critical to the success of your QMS. (It is recommended to have more than one auditor to ensure no one is auditing his or her area of responsibilities). The internal auditor must be independent of the area being audited to ensure objective results. Internal audits have many benefits including preparing your organization for external audits. Internal audits are audits that are performed by your organization and are a self-examination of your organization’s QMS, performed on-site. ISO 13485 PowerPoint Training Materials.ISO 13485 Online Training Course Listing.ISO 13485 Internal Auditor Training Courses.ISO 13485 Lead Auditor Training Courses.Simple Steps to ISO 13485 Certification Follow our proven and manageable step-by-step process for a successful ISO implementation project. 
Quality Audit requirements according FDA Quality System Regulation are reported in the section 21 CFR 820.22. The text of the section is reported below:Įach manufacturer shall establish procedures for quality audits and conduct such audits to assure that the quality system is in compliance with the established quality system requirements and to determine the effectiveness of the quality system. Quality audits shall be conducted by individuals who do not have direct responsibility for the matters being audited. Corrective action(s), including a reaudit of deficient matters, shall be taken when necessary. The dates and results of quality audits and reaudits shall be documented.īased on this text of the regulation it is possible to basically summarise the key requirements for the internal audit process: A report of the results of each quality audit, and reaudit(s) where taken, shall be made and such reports shall be reviewed by management having responsibility for the matters audited. Quality Audits shall be performed to ensure compliance of the Quality System to the applicable regulatory requirements.Independence of the auditors shall always be demonstrated. 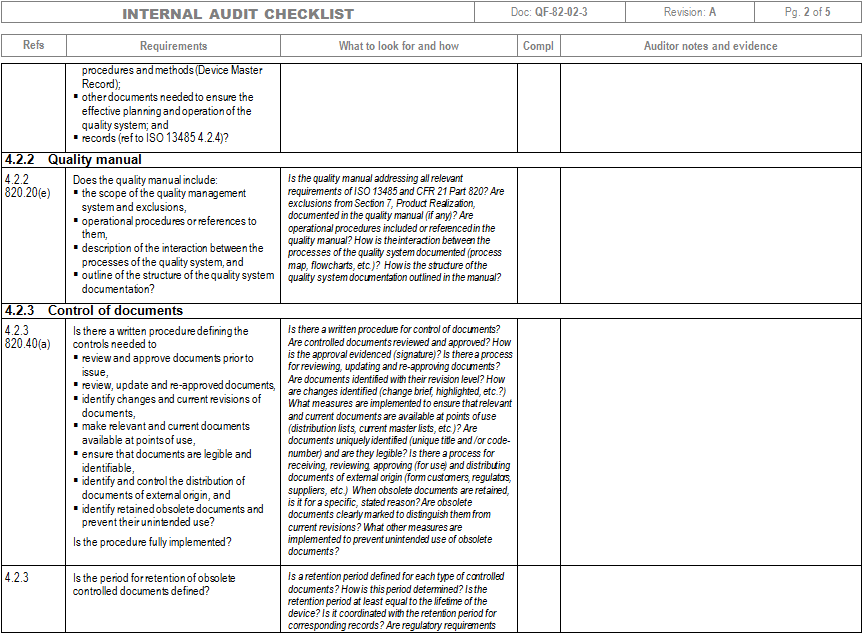
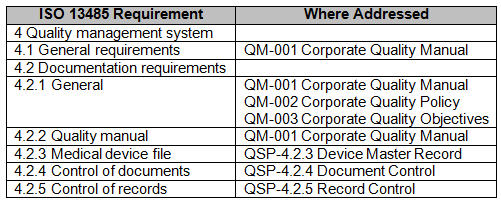
The link between Internal Audits and CAPA process shall be properly demonstrated and documented.Internal Audit Plan and reports shall be documented.Internal Audit Requirements According to ISO 13485:2016 
The section related to internal audit within ISO 13485:2016 is the clause 8.2. There are mainly the same requirements already explained in the previous section related to FDA Quality System Regulation. However, in addition to what has already be mentioned in the previous paragraph, there are some additional requirements which is important to mention. Specifically, one of the most important requirement is the risk-based approach in the planning of the internal audits. In fact, ISO 13485 requires internal audits to be planned “ taking into consideration the status and importance of the processes and area to be audited, as well as the results of previous audits“. This is very important and it can be included in the framework of those requirements that go in the direction of the implementation of risk management principles in the Quality Management System of an organization. It is very important to document an audit plan which takes in consideration the internal audits to be performed through the whole year. In the audit plan, the specific processes to be audited need to be mentioned along with the timing on when that specific audit is planned. In fact, the text of the ISO standard reports: Moreover, it definitely makes sense to include the specific audit criteria for each specific process to be audited.Īlso within ISO 13485 there is the requirements associated to the link between the internal audit process and the CAPA process. The management responsible for the area being audited shall ensure that any necessary corrections and corrective actions are taken without undue delay to eliminate detected nonconformities and their causes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
